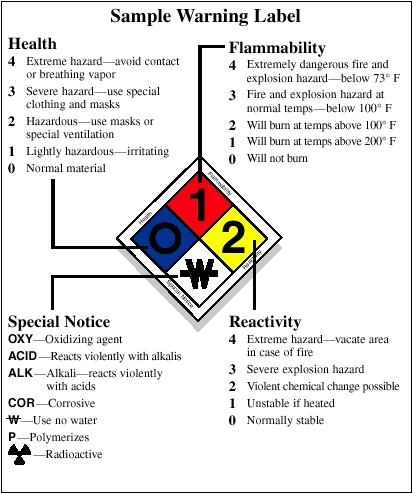
43 drug warning labels examples
CFR - Code of Federal Regulations Title 21 - Food and Drug Administration § 201.312 - Magnesium sulfate heptahydrate; label declaration on drug products. § 201.313 - Estradiol labeling. § 201.314 - Labeling of drug preparations containing salicylates. § 201.315 - Over-the-counter drugs for minor sore throats; suggested warning. § 201.316 - Drugs with thyroid hormone activity for human use; required warning. Academic Journals | American Marketing Association Journal of Marketing (JM) develops and disseminates knowledge about real-world marketing questions useful to scholars, educators, managers, policy makers, consumers, and other societal stakeholders around the world.It is the premier outlet for substantive marketing scholarship. Since its founding in 1936, JM has played a significant role in shaping the content and boundaries of …
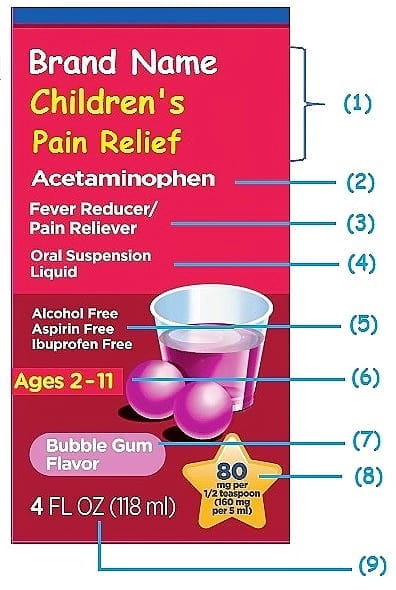
What Information Should Be on Drug Labels? - MedicineNet Before any drug can be legally sold, it must be assigned a Drug Identification Number (DIN). The FDA recommends manufacturers to provide data demonstrating product safety, effectiveness in meeting label claims, potency, and purity. When the manufacturer meets these requirements, the FDA assigns the drug a registration number.

Drug warning labels examples
Prop 65 Labeling Requirements With Examples - Coast Label Company In order to comply with the law, Prop 65 labels need the following elements: A warning symbol. The word " WARNING " (bolded, in all caps) An explicit statement of the chemical causing exposure. A statement about whether the chemical could cause cancer, birth defects, or reproductive harm. Some products are exempt from labeling requirements. › nutritionsource › food-labelUnderstanding Food Labels | The Nutrition Source | Harvard T ... Chile implemented the Law of Food Labeling and Advertising in 2016, comprised of mandatory front-of-package (FOP) warning labels, restrictions on child-directed marketing, and the banning of sales in schools of all foods and beverages containing added sugars, sodium, or saturated fats that exceeded set nutrient or calorie thresholds. [1] en.wikipedia.org › wiki › Boxed_warningBoxed warning - Wikipedia The FDA can require a pharmaceutical company to place a boxed warning on the labeling of a prescription drug, or in literature describing it. It is the strongest warning that the FDA requires, and signifies that medical studies indicate that the drug carries a significant risk of serious or even life-threatening adverse effects.
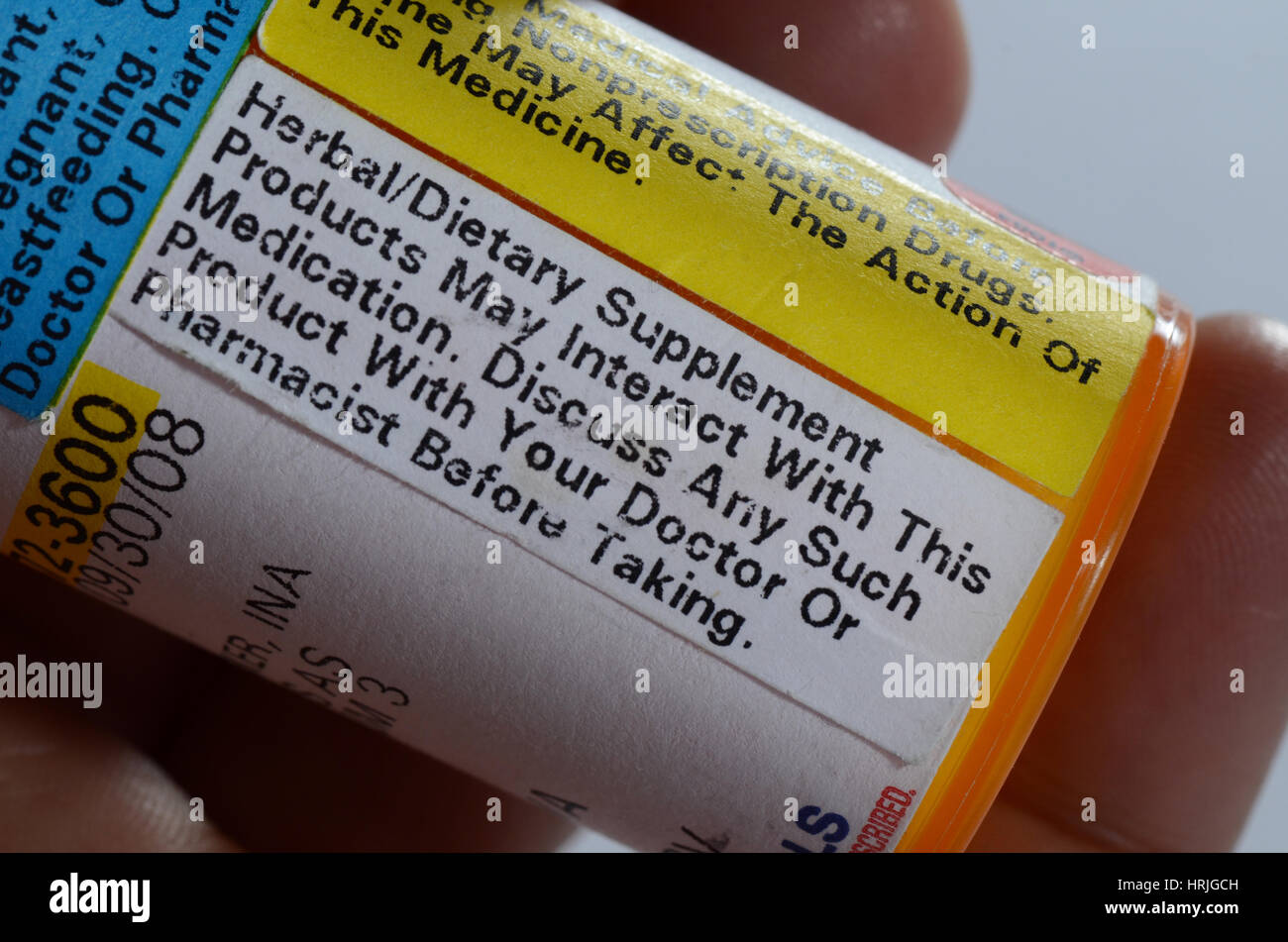
Drug warning labels examples. FDA Drug Safety Communication: FDA strengthens warning … The U.S. Food and Drug Administration (FDA) is strengthening an existing label warning that non-aspirin nonsteroidal anti-inflammatory drugs (NSAIDs) increase the chance of a heart attack or stroke. Misunderstanding Drug Warning Labels - Medscape The example above also highlights a cognitive process that is common among individuals with low literacy skills. ... Prescription Drug Warning Labels and Respondent Interpretations ( n = 74) Label How To Read Drug Labels: A Step by Step Guide for Consumers However, the following steps will help you make sense of everything. 1. Start with the basics. Make sure you can identify the following information on the label: Name of the active ingredient in the medication. Health conditions that the drug treats. Strength, meaning how much active ingredient is in each dose. How to Read Food Labels Without Being Tricked - Healthline 19.08.2020 · Nutrition labels can be confusing and misleading. This article sets the record straight about how to avoid falling into some of these consumer traps.
CBD Labeling Requirements and Guidelines - Avery The warning letter stated the CBD company is in violation for using false claims on their product labels and website. "Based on the inspection and a review of your product labels and your websites, we have identified serious violations of the Federal Food, Drug, and Cosmetic Act (the Act) and applicable regulations," the FDA said in the letter. Medication Guides | FDA - U.S. Food and Drug Administration FDA requires that Medication Guides be issued with certain prescribed drugs and biological products when the Agency determines that: certain information is necessary to prevent serious adverse ... Black Box Warning - StatPearls - NCBI Bookshelf Boxed warnings (formerly known as Black Box Warnings) are the highest safety-related warning that medications can have assigned by the Food and Drug Administration. These warnings are intended to bring the consumer's attention to the major risks of the drug. Medications can have a boxed warning added, taken away, or updated throughout their tenure on the market. Over 400 different ... Drug Alerts and Statements | FDA FDA Drug Alerts and Statements ... FDA approves label changes to ... 8/12/2016 FDA finds drugs under investigation in the U.S. related to French BIA 10-2474 drug do not pose similar safety ...
› ama-academic-journalsAcademic Journals | American Marketing Association Journal of Interactive Marketing aims to identify issues and frame ideas associated with the rapidly expanding field of interactive marketing, which includes both online and offline topics related to the analysis, targeting, and service of individual customers. Delay in Graphic Warning Labels on Cigarettes Cost Lives: Study The U.S. Food and Drug Administration has a set of 13 graphic warning labels that would cover half of a cigarette pack's front and back. The warnings "stand to represent the most significant change to cigarette labels in 35 years," the agency says. The labels contain disturbing images and short messages about lesser-known risks of cigarette ... FDALabel: Full-Text Search of Drug Product Labeling | FDA For example, recent publications describe how information in drug labeling can be used to aid and facilitate drug repurposing 4 as well as applications in precision medicine, drug safety, and ... › article › grapefruit-drugCommon Grapefruit Juice Drug Interactions - Drugs.com May 18, 2022 · While grapefruit is a nutritious fruit, many patients are concerned about the potential for drug interactions with grapefruit juice. Maybe you've receive a medication prescription container with an affixed warning label that recommends you avoid grapefruit or grapefruit juice while taking the medication.
FDA's Labeling Resources for Human Prescription Drugs | FDA Human prescription drug labeling (1) contains a summary of the essential scientific information needed for the safe and effective use of the drug; and (2) includes the Prescribing Information, FDA ...
Misunderstanding Drug Warning Labels - Medscape Results: Among this sample of patients with low literacy skills, rates of correct interpretation for the eight warning labels ranged from 0% to 78.7%. With the exception of the most basic label ...
Examples of Metaphors in Literature - ProWritingAid 14.01.2022 · She hissed her warning— “Keep your mouth shut or I’ll shut it for you.” In this case, no snake is mentioned, but the “hiss” implies the comparison of “she” to that dangerous, threatening creature. Visual Metaphor. Visual metaphors show, …
Introduction to the New Prescription Drug Labeling by the FDA Example of highlights section (for a fictitious drug). ... Many drug product labels contain a boxed warning, often referred to by health care practitioners as a black-box warning, to emphasize ...
How to Write a Warning Label to Avoid Product Liability The Warnings Should Be Prominent, Clear and Understandable A warning is only effective if people see and understand it. If the purpose of the warning is to avoid product liability, placing the warning on the last page at the bottom of the instruction manual in a tiny font is counterproductive. The warning should be printed prominently in the ...
Implementation Guidelines for Alcohol and Drug Regulations DOT's final rule, 49 CFR part 40, "Procedures for Transportation Workplace Drug and Alcohol Testing Programs," took effect August 1, 2000. The rule modified and expanded the procedural requirements for controlled substances testing, including specimen collection procedures, laboratory testing procedures, medical review officer (MRO) procedures, and substance abuse
Understanding Food Labels | The Nutrition Source | Harvard T.H. All FOP labels in the U.S. are voluntary, which allows food manufacturers to highlight or hide the nutrition information they choose to help promote or preserve sales. If warning labels became mandatory, as public health advocates propose, the pressure on manufacturers would increase to change certain products to improve their nutritional quality.
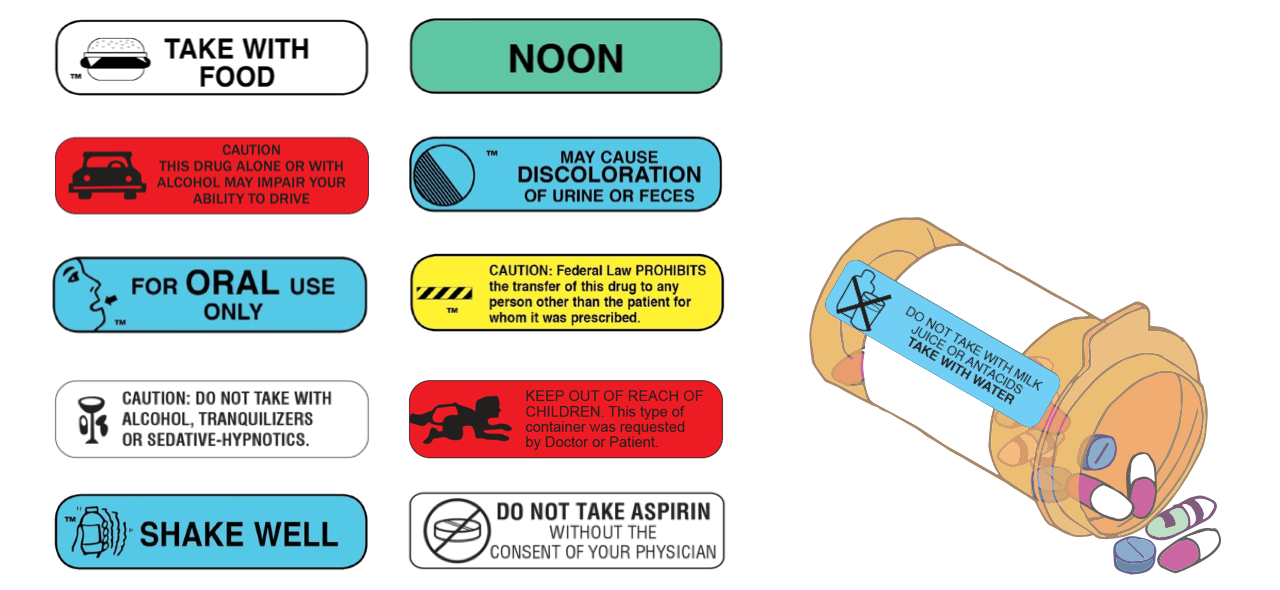
What are Auxiliary Labels? - PTCB Test Prep Definition: "auxiliary labels are cautionary labels added to a dispensed medicine to provide extra information to the patient on the safe administration, use, and storage of their medicines". In other words, auxiliary labels refer to important features of the medicine that patients must keep in mind. Examples of common auxiliary labels include:
Warning label - Wikipedia Warning labels have been produced for different items. In some cases, rumors have developed of labels warning against some very strange occurrences, such as the legendary microwave warning that allegedly states 'do not dry pets in microwave'. Some companies hold 'strange warning label competitions' such as the former M-law wacky warning labels ...
Table of Pharmacogenomic Biomarkers in Drug Labeling | FDA Drug Therapeutic Area* Biomarker † Labeling Sections Abacavir: Infectious Diseases: HLA-B: Boxed Warning, Dosage and Administration, Contraindications, Warnings and Precautions
CFR - Code of Federal Regulations Title 21 - Food and Drug Administration For the most up-to-date version of CFR Title 21, go to the Electronic Code of Federal Regulations (eCFR). Sec. 203.38 Sample lot or control numbers; labeling of sample units. (a) Lot or control number required on drug sample labeling and sample unit label. The manufacturer or authorized distributor of record of a drug sample shall include on ...
› inspections-compliance-enforcementMercola.com, LLC - 607133 - 02/18/2021 | FDA Mar 04, 2021 · WARNING LETTER. Date: February 18, 2021. Cc: TAHarrison@Venable.com LGordon@venable.com Venable LLP Attn: Todd A. Harrison Attn: Leonard Gordon 600 Massachusetts Ave., NW
List of Black Box Warnings for Pharmacists | PharmaFactz When a black-box warning is issued, it informs healthcare providers and prescribers of serious adverse effects of specific drugs and enhances their clinical judgment. For example: when atypical antipsychotics were assigned a black box warning for use in patients with dementia (as it increases the risk of death) - prescription use of ...
Boxed warning - Wikipedia In the United States, a boxed warning (sometimes "black box warning", colloquially) is a type of warning that appears on the package insert for certain prescription drugs, so called because the U.S. Food and Drug Administration specifies that it is formatted with a 'box' or border around the text. The FDA can require a pharmaceutical company to place a boxed warning on the …
Drug Interactions: What You Should Know | FDA Further, drug labels may change as new information becomes known. That's why it's especially important to read the label every time you use a drug. The " Active Ingredients " and " Purpose ...
en.wikipedia.org › wiki › Warning_labelWarning label - Wikipedia Warning labels have been produced for different items. In some cases, rumors have developed of labels warning against some very strange occurrences, such as the legendary microwave warning that allegedly states 'do not dry pets in microwave'. Some companies hold 'strange warning label competitions' such as the former M-law wacky warning labels ...
Warning Label Accident Statistics | LegalMatch A warning defect, or a warning label defect, is a specific type of legal theory that is most commonly used as the basis of defective products liability lawsuits. Simply put, a warning defect can occur when the manufacturer of a product fails to include sufficient warnings associated with the product. This is generally in the form of a warning ...
› drugs › resources-you-drugsDrug Interactions: What You Should Know | FDA Over-the-counter (OTC) drug labels contain information about ingredients, uses, warnings and directions that is important to read and understand. The label also includes important information ...
en.wikipedia.org › wiki › Boxed_warningBoxed warning - Wikipedia The FDA can require a pharmaceutical company to place a boxed warning on the labeling of a prescription drug, or in literature describing it. It is the strongest warning that the FDA requires, and signifies that medical studies indicate that the drug carries a significant risk of serious or even life-threatening adverse effects.
› nutritionsource › food-labelUnderstanding Food Labels | The Nutrition Source | Harvard T ... Chile implemented the Law of Food Labeling and Advertising in 2016, comprised of mandatory front-of-package (FOP) warning labels, restrictions on child-directed marketing, and the banning of sales in schools of all foods and beverages containing added sugars, sodium, or saturated fats that exceeded set nutrient or calorie thresholds. [1]
Prop 65 Labeling Requirements With Examples - Coast Label Company In order to comply with the law, Prop 65 labels need the following elements: A warning symbol. The word " WARNING " (bolded, in all caps) An explicit statement of the chemical causing exposure. A statement about whether the chemical could cause cancer, birth defects, or reproductive harm. Some products are exempt from labeling requirements.
































Post a Comment for "43 drug warning labels examples"